Numerous problems have been reported due to pollution deposition on the surface of high voltage outdoor insulators. Although accumulation of pollution is typically most severe in coastal and desert areas, industrial regions and urban areas, almost all outdoor insulators eventually become contaminated after several years’ service. When such contaminated surfaces are exposed to wetting from rain, fog or dew, dissolved pollution creates a conductive electrolytic layer. This is followed by formation of dry-band arcing that can profoundly affect insulator pollution flashover behaviour. Hence, performance of insulation materials in polluted areas must be carefully addressed and the problem of flashover of contaminated insulators needs to be resolved.
High temperature vulcanized silicone high voltage insulators are known for exceptional hydrophobic properties such that water droplets stand separately over the surface and do not form a continuous film.
This edited past contribution to INMR by the Dept. of Applied Sciences at the University of Quebec in co-operation with K-Line Insulators reviewed opportunities to further improve pollution flashover behaviour.
Listen to Online Lecture on Super-Hydrophobic Silicone for High Voltage Insulators by A.J. Carreira
Super-hydrophobic surfaces offer a water contact angle (WCA) >150˚ and a contact angle hysteresis (CAH) or sliding angle (SA) <10˚. This feature has attracted significant attention in a wide range of applications requiring anti-corrosive, ice-phobic, anti-biofouling, non-wetting and self-cleaning surfaces. Inspired by the unique self-cleaning of lotus leaves, also known as the “lotus effect”, this property is being widely used in industrial applications. Since pollution particles are generally hydrophilic, they tend to adhere to water droplets rather than to a super-hydrophobic, self-cleaning surface.
Two main approaches are used to produce a super-hydrophobic surface: 1. roughening the surface of a material with low surface energy and 2. depositing a low surface energy material on an already rough surface. Silicone rubber is a low surface energy material and it can be roughened sufficiently to produce a super-hydrophobic surface. A self-cleaning silicone rubber surface is capable of repelling water droplets and thereby removing contaminant particles that adhere to it, i.e. any dust and pollution is easily removed as water droplets roll off its super-hydrophobic surface. Therefore, fabrication of a super-hydrophobic surface having self-cleaning properties can effectively address issues associated with contaminated insulator surfaces.
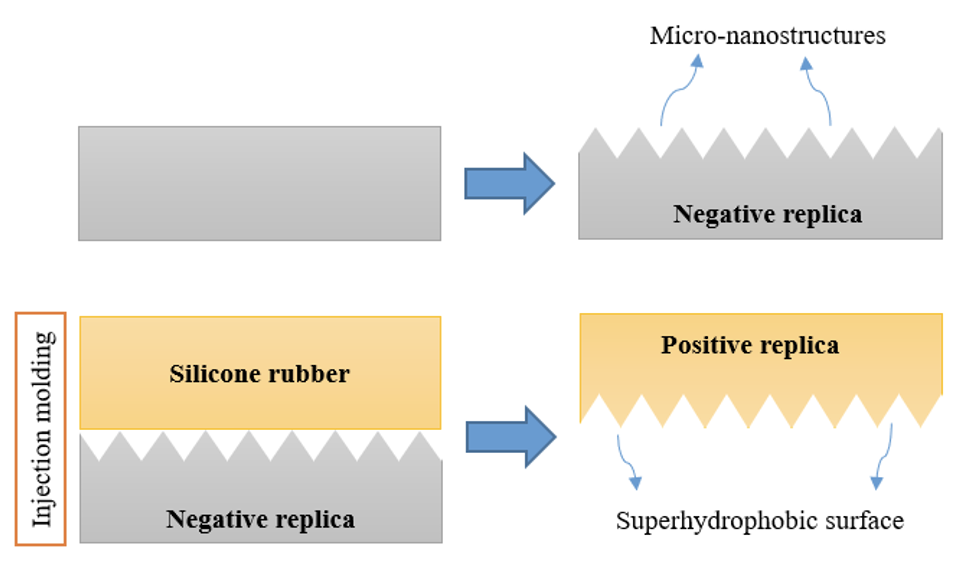
Although several methods are available to produce super-hydrophobic surfaces, direct replication was selected since it is simple, efficient and inexpensive and can also easily be industrialized. Using this method, the surface does not undergo chemical changes. Rather, an appropriate micro-nanostructure is created on the surface of a negative replica. A positive replica is then produced by means of injection molding (see Fig. 1). The super-hydrophobic silicone rubber surfaces thus created demonstrate ultra water repellence as well as self-cleaning properties under different contamination conditions.
Assessment of Super-Hydrophobic Properties
The smooth sample of silicone rubber originally had a WCA ~116º ± 2º and CAH >30º (see Fig. 2a & c) whereas the new replicated silicone surface showed a WCA ~161º ± 1.3º and CAH ~7º (see Fig. 2b & d). Due to the low CAH, water droplets on the super-hydrophobic surface easily rolled off, demonstrating a self-cleaning capability.

Micro-nanostructures on the silicone rubber surface allow achieving such high WCA and low CAH. Creation of such micro-nanostructures after the direct replication process on the silicone surface has been studied using SEM analysis (see Fig. 3).

Ultra Water Repellent Silicone Rubber Surfaces
To demonstrate super-hydrophobic properties, a sample of micro-nanostructured silicone rubber surface was immersed in a water-filled Petri dish. The super-hydrophobic silicone surface remained dry while the height of the water reached up to ~3 mm (the so-called Moses Effect). The blue water droplets demonstrate dryness of the surface.

Water jet tests serve to confirm ultra water repellent properties of a surface. Here, the water jet was applied by a syringe equipped with a needle, under normal force, on both hydrophobic and super-hydrophobic samples placed on a flat surface. When the jet was sprayed on the smooth silicone rubber, water accumulated on its surface (see Fig. 5a).
But when a water jet was applied to the super-hydrophobic silicone rubber surface, it rebounded (see Fig. 5b). This demonstrates that a current of high-pressure water cannot remain on the surface of micro-nanostructures, i.e. the super-hydrophobicity of the manufactured surface is stable.

The behaviour of a surface exposed to continuous impact of water droplets is representative of water repellence and this behaviour can be perceived when surfaces are exposed to rainfall. In such a test, water droplets of certain diameter are released from a height and impact silicone rubber surfaces placed on 10˚ tilted stand. As seen in Fig. 6a, two consecutive water droplets adhered to the smooth silicone rubber surface upon impact.
However, water droplets rebounded off the super-hydrophobic silicone rubber surface without leaving a residue (Fig. 6b). It is worth noting that formation of a secondary or satellite droplet was also observed in the case of the manufactured super-hydrophobic surface and further emphasizes its ultra-water-repellent property.

Self-Cleaning Silicone Rubber Surfaces
A super-hydrophobic silicone rubber surface with ultra water repellent properties and low CAH is self-cleaning. This self-cleaning property was compared to that of the smooth normal silicone rubber surface by adding the same amount of fine kaolin particles as contaminants to each surface.
As seen in Fig. 7a, a water film forms on the smooth surface with kaolin. By contrast, water droplets readily clean the surface of the super-hydrophobic silicone rubber along their flow path. As seen in Fig. 7b, only several water droplets are sufficient to clean the surface.

A dirty multi-component suspension of SiO2 particles, salt, carbon black and glycerine was added to water to evaluate functionality of a super-hydrophobic surface under polluted wet conditions. The hydrophobic and super-hydrophobic samples were immersed in equal amounts of suspension and then maintained at ambient temperature of 70˚C for 2 h. Figs. 8a and b show the surface appearance of each sample after water evaporation. While the surface of the smooth silicone sample is completely covered by the dirty sediment, most of the super-hydrophobic silicone rubber surface remains clean.
After cleaning each surface using the same amount of de-ionized water, it was observed that water accumulated on the smooth surface, forming a continuous film (see Fig. 8c). However, the surface of the super-hydrophobic surface readily cleaned off remaining dirt sediments. Such remarkable self-cleaning behaviour of super-hydrophobic surfaces is highly desirable when insulation materials are being used in highly polluted service areas.

Conclusions
Micro-nanostructured silicone rubber surfaces were manufactured by direct replication using an injection molding process with the goal of helping overcome insulation pollution flashovers. These silicone rubber surfaces showed super-hydrophobic properties, i.e. a WCA >150˚ and CAH <10˚, and also demonstrated extraordinary water repellence, confirmed by immersion in water testing as well as water jet impact and water impact tests.
These experiments were beneficial in investigating functionality of super-hydrophobic silicone rubber surfaces exposed to rainfall. Manufactured surfaces also demonstrated self-cleaning properties due to their low CAH. Kaolin powders were easily removed from super-hydrophobic surfaces using water droplets and super-hydrophobic surfaces showed excellent self-cleaning properties when exposed to dirty, wet conditions.
Although they accumulated only negligible residue on their surface, the dirt residue was easily cleaned using several water drops. This confirms that super-hydrophobic surfaces can be considered a highly effective option to reduce risk of pollution flashover of insulation in highly polluted service areas.
References
[1] http://www.inmr.com/pollution-flashover-insulators/
[2] Maghsoudi, K., Momen, G., Jafari, R., Farzaneh, M., & Carreira, T. (2018, October). Micro-Nanostructured Silicone Surfaces for High-voltage Application. In 2018 IEEE Conference on Electrical Insulation and Dielectric Phenomena (CEIDP) (pp. 179-182). IEEE.
[3] Gençoğlu, M. T., & Cebeci, M. (2008). The pollution flashover on high voltage insulators. Electric Power Systems Research, 78(11), 1914-1921.