Several years ago, service inspections of composite station post insulators by utilities in Europe revealed instances of corona occurring not only close to the flange but also at some distance from the high voltage end. This finding was unexpected given that corona activity is usually linked to high electric field. Thermal inspection of these insulators detected hot spots in the same places where corona had been observed. Close visual inspection then revealed multiple punctures along the housing in those locations where corona and hot spots had been detected. Follow-up laboratory investigation found that the punctured areas were all marked by poor adhesion between the core rod and the polymeric housing. This suggested that one of the ways composite insulators can deteriorate and fail is from manufacturing processes that do not ensure proper adhesion along this critical interface.

This edited past contribution to INMR by experts at Wacker Silicones reviewed processes to achieve optimal adhesion between silicone elastomer and a substrate.
The different types of elastomers grouped in International Standard ISO 1629,4 are coded using a single letter, the most important of which are:
M: Rubbers having saturated carbon chain of the polymethylene type;
O: Rubbers having carbon and oxygen in the polymer chain; and
Q: Rubbers having silicon and oxygen in the polymer chain;
According to this standard, silicone elastomers are then defined by adding the substituent group on the polymer chain prior to the Q designation.thus leading to the following main groups:
MQ or polydimethylsiloxane (PDMS) that describes a polymer in which both side groups ‘R’ are methyl groups;
VMQ that describes a PDMS polymer in which small numbers of methyl groups have been substituted by vinyl groups;
PVMQ that stands for PDMS with small amount of methyl groups exchanged by phenyl groups; and
FVMQ that describes PDMS in which small numbers of methyl groups have been replaced by trifluoropropyl groups.
The silicone material used in medium and high voltage insulation applications belongs to VMQ silicone elastomers, which consist mainly of the silicone polymer described above along with reinforcing filler. Separation into different product groups among these elastomers is then made according to the different viscosities of the formulation as well as the cross-linking mechanism used. For example, 3 distinct groups are used in T&D applications: high consistency rubber (HCR), liquid silicone rubber (LSR) and room temperature vulcanizing (RTV) rubber. HCR products rely on both peroxide curing and platinum catalyst addition curing. Moreover, one can find addition cured LSR and RTV-2 formulations as well as condensation cured RTV-1 systems.
Adhesion Technologies
Adhesion of silicone elastomers to different substrates is an important topic for a wide array of applications. Composite materials, such as insulators, consisting of silicone rubber and substrates such as a core rod or tube can be achieved using different processing methodologies.
In rare cases, adhesion on a substrate can be achieved with standard materials in a regular production process. However, achieving good adhesion in composite materials generally requires special effort and methods that include:
• Application of primers;
• Surface treatment;
• Composite design with undercuts; and
• Use of self-adhesive grades of silicone rubber (not feasible for T&D applications).
For technical reasons, the technology of self-adhesive silicone rubber grades is not feasible for T&D applications and not discussed here.
To generate build-up of good adhesion between a substrate and a silicone elastomer using over-moulding, a complex interaction of physical and chemical processes must take place. Influencing factors include functional groups on the surface of the substrate and silicone, surface energy and operating temperature. A high surface energy with the presence of reactive chemical groups is necessary to achieve good bonding. For example, surface treatment with plasma, corona, Pyrosil® or flame treatment can generate these desired conditions but with relatively high investment in processing technology. Plasma, corona and flame treatment all generate reactive chemical groups on the surface of the substrate by applying energy with ionic plasma, corona discharge or natural gas flame respectively. By contrast, Pyrosil® technology is based on bringing new reactive groups to the surface by deposition of combustion chemical vapour (see Fig. 1).

The gas flame is enriched with a precursor, an organo-silicon compound, that is burned off during the process. The ash generated deposits as an amorphous silicate on the surface and this thin layer will form a strongly adhering coating that ensures good adhesion. Using a chemical primer can also establish chemical bonding between silicone rubber and substrate (see Fig. 2). Ensuring stable and reproducible results requires ensuring that all parameters necessary for the chemical reaction of the primer are properly followed.
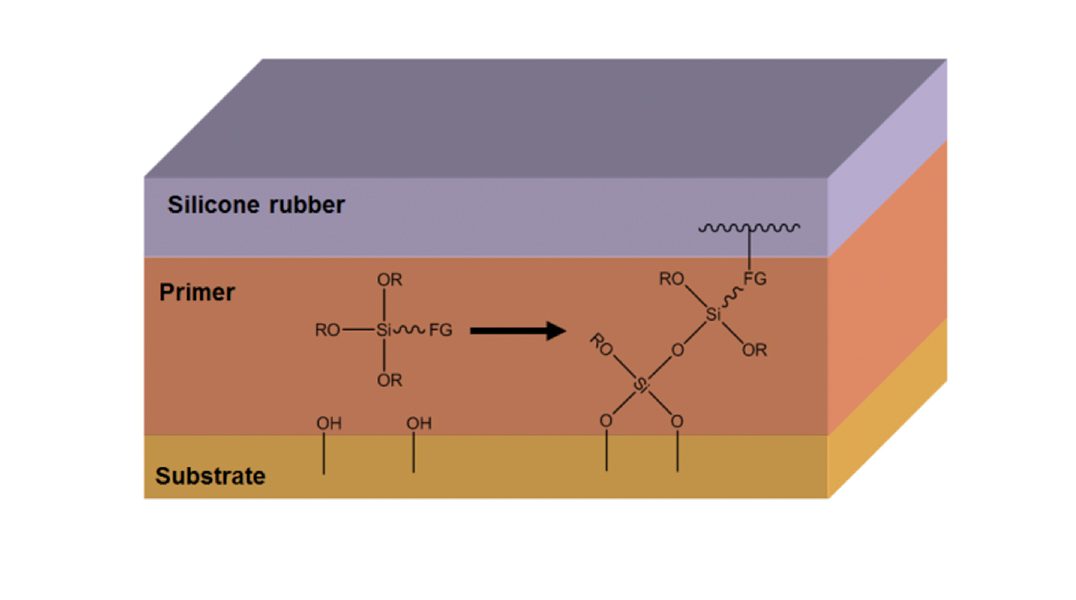
The primer layer is typically applied by means of spraying, brushing or dipping. After ‘flash-off’ of the solvent, the primer is cured with humidity from the ambient air. In some cases, baking the substrate with the cured primer layer improves adhesion. Finally, the silicone is cured onto the substrate by compression, transfer or injection moulding.
It is important to note that besides parameters like humidity and temperature other major influencing factors are decisive for a good result. Based on the material combination of substrate, primer and silicone rubber the adhesion force will decrease over storage time of the primered part. Furthermore it is important to know the surface conditions of the substrate. In general a rough surface offers more surface area for establishing a chemical bonding. In this case it may also be common thinking that applying more primer will improve the situation. This is a completely wrong approach. A thick layer of primer levels out the surface roughness of the substrate and reduces the contact surface on the one hand and on the other hand builds up a thick layer of a resin based, mechanically less stable respectively brittle material which breaks easy (see Fig. 3).
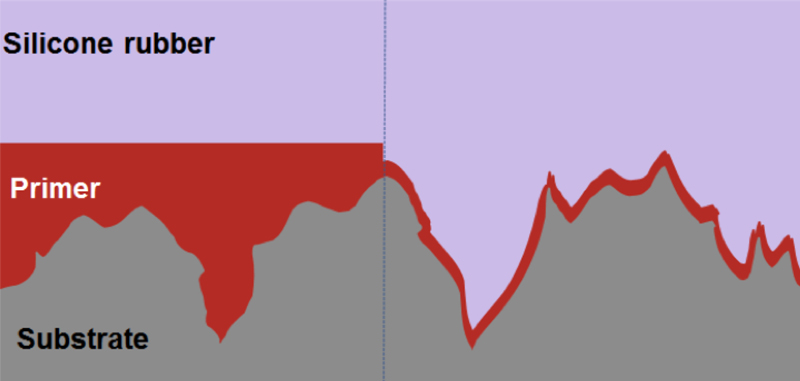
Appropriate preparation and cleaning of surfaces is crucial for successful application of adhesion promoter. Non-polar polymers or polluted surfaces can prevent a sufficient wetting of the entire surface and reduce therefore the adhesive surface.
Chemical adhesion primers can achieve very good result in chemical bonding of silicone rubbers to various substrates if the processing complies with the optimum chemical process and parameters for the individual material combination.
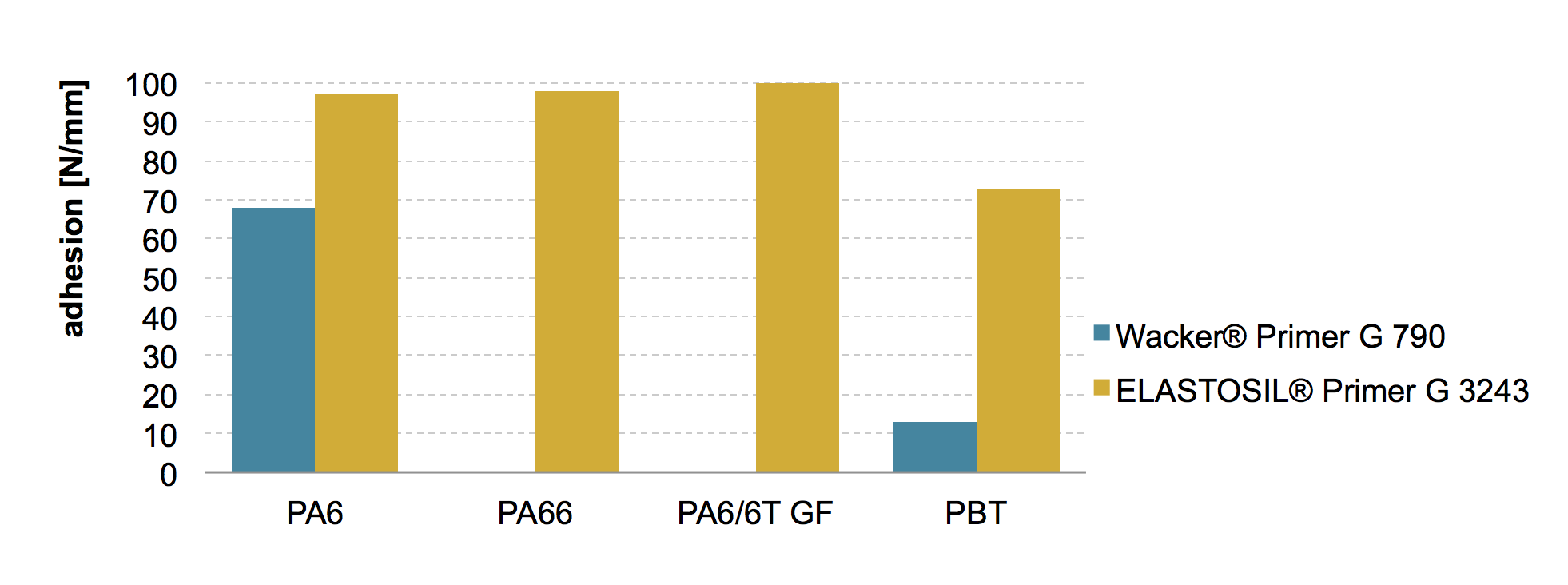
In order to accommodate this different primers for the individual crosslinking systems are available. Those are designed to give optimal adhesion respectively chemical bonding to silicone in respect to the crosslinking system. Cross trials using addition and peroxide curing standard silicone with PA and PBT substrates and primer designed for addition respectively peroxide cured silicones showed this effect significantly. The results of the peroxide curing ELASTOSIL® R 401/60 on substrates either pretreated with Wacker® Primer G 790 recommended for addition curing products and Primer ELASTOSIL® G 3243 recommend for peroxide curing formulations. It is very obvious that the appropriate primer needs to be used to gain an optimum adhesion (Fig. 4). Typical good adhesion force values given at for these internal adhesion tests are above 10 N/mm.
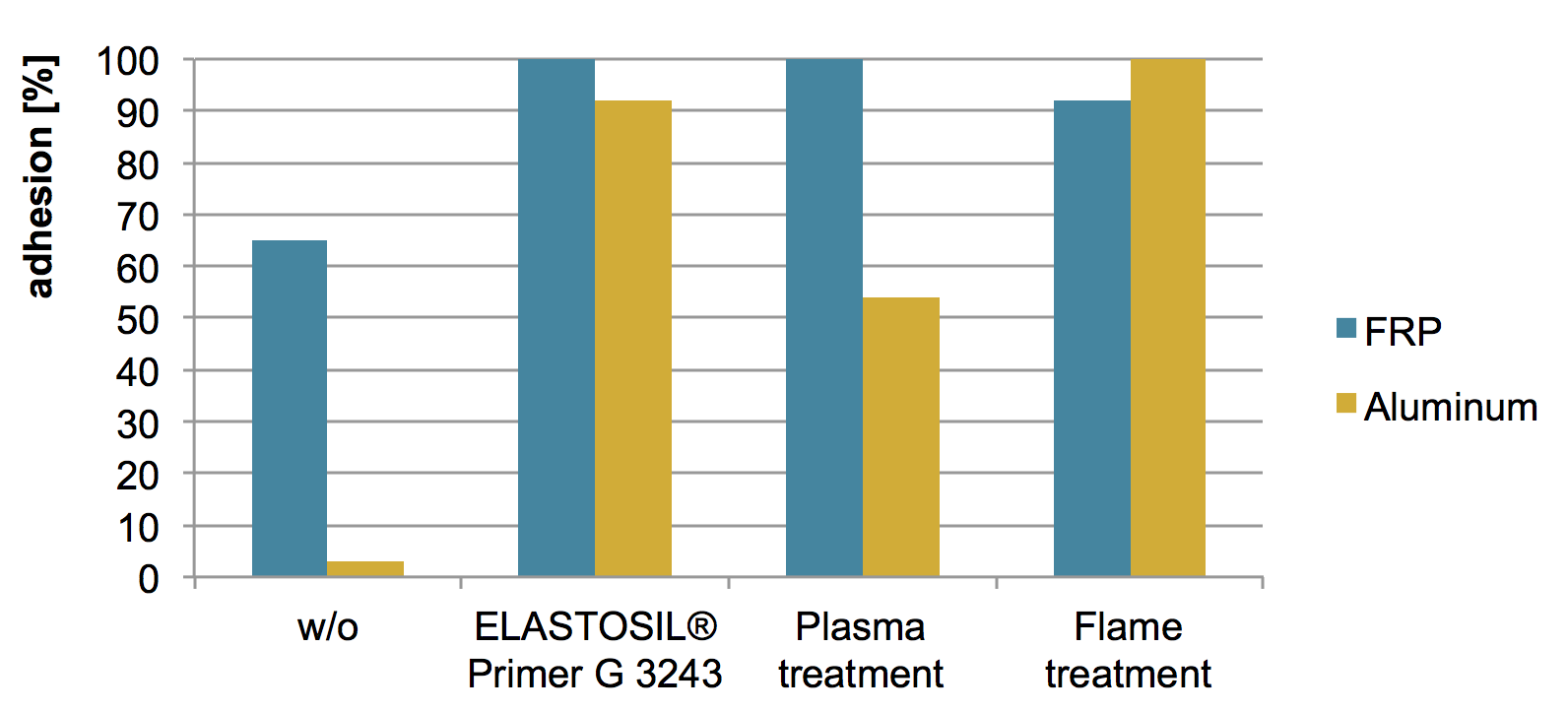
The comparison of the different physical and the chemical pretreatments of T&D relevant substrates like FRP or Aluminum has been performed using a POWERSIL® 310 formulation. As this is a peroxide curing formulation the ELASTOSIL® Primer G 3243 has been used. In the given chart (Fig. 5) an increase of adhesion force using the pretreated FRP compared to the untreated one is visible. Even stronger is the effect on Aluminum.
The right choice and individual combination of methods mentioned in this article display a broad variety to achieve the best possible solution for any adhesion problem. By physical and chemical surface treatment basically any substrate can be activated for good and stable adhesion.
References
[1] DIN ISO 1629, Rubber and lattices —Nomenclature, 2013.
[2] J. Lambrecht, Silicone Technology Review, INMR Issue 111, 2016
[3] ASTM D 1418, Standard Practice for Rubber and Rubber Latices —Nomenclature, 2015.
[4] Pachaly, B., Achenbach, F., Herzig, C., Mautner, K., Silicones, Winnacker/Küchler, Chemische Technik: Prozesse und Produkte, Band 5: Organische Zwischenverbindungen, Polymere, 2005.
[5] Tomanek, A., Silicones & Industry – a compendium for practical use, instruction and reference, Wacker Chemie AG, Carl Hanser Verlag, München, 1991.
[6] Sura Instruments GmbH, Das Pyrosil®-Verfahren, 2004.